| |
|
 |
|
Microscope setup (with incubator) for Confocal 3D Optical Sectioning
Capturing high quality images of cells and molecular events at high speed
- Galvano scanner for high resolutions (16 Megapixels) and high speed (10fps) scanning
- Ultrafast (420fps) scanning with resonant scanner
- Stimulation + Imaging with hybrid scanner
Multicolour Imaging, Spectral Imaging, Simultaneous photoactivation and imaging, photoactivation dye, FRAP, FRET, Caged Compound, Calcium imaging dye
| Multicolour Imaging |
|
|
- 4 Colour Fluorescence + DIC
- Simultaneuos/Custom Sequential excitation
|
< Zebrafish labeled with 4 probes. (From A1+ Brochure)
Nucleus (Blue): Hoechst33342, Pupil (Green): GFP,
Nerve (Y): Alexa555, Muscle (Red): Alexa647. |
| |
|
| Spectral Imaging |
|
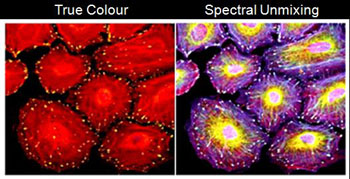 |
- One shot 32 channels
- Seperate overlapping fluorescence spectra
- Elimintating autofluorescence
|
| <Spectral and unmixed image of 5 probes labeled HeLa cells (From A1+ Brochure) |
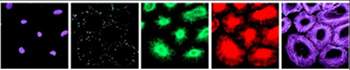 |
<Unmixing result.
From left: Nucleus(DAPI), Vinculin(Alexa488), Vimentin(Alexa568), Tubulin(Alexa594), Actin (Alexa633) |
| |
|
|
|
|
| |
|
 |
|
Microscope setup for Super-resolution N-STORM, TIRF and Live Cell Imaging
- N-STORM: 10X better resolution than conventional microscopy
- TIRF: Study of cell event close to plasma membrane
- Live Cell: Equipped with on-stage incubator, shutter and filter wheel for live cell imaging
| N-STORM (STochastic Optical Reconstruction Microscope) |
- Super-resolution image with localization of each fluorophore
- Multichannel 2D/3D (3 colours)
- Resolution XY:~ 20nm; Z: ~50nm
- N-STORM dyes recommended
- Continuous mode available
- Contact us for sample preparation protocol and enquiries
|
| |
2 Colours N-STORM : |
Red-Microtubules (Cy3-Alexa647) |
| |
Green- Mitochondria (Alexa405-Alexa647) |
|
| |
| |
| TIRF (Total Internal Reflection Fluorescence) |
|
- Excites fluorophores within 100nm above coverslip
- Suitable for observation events occur close to plasma membrane
- 4 Colours TIRF
|
| |
Cells immunocytochemically labeled for the protein tubulin under widefield and TIRF observation
(Courtesy of MicroscopyU.com) |
|
|
|
|
|
| |
|
 |
Microscope for slide viewing: Brightfield, Darkfield, Phase contrast, DIC and Fluorescence |
| |
Large image stitching of HE staining slide under 10X objective |
|
| |
Brightfield and Darkfield observation of mouse blood (40X objective) |
|
|
| |
AZ100: Multizoom Microscope |
 |
|
|
| |
| Configuration: Brightfield, DIC and Fluorescence |
| Magnification: 5X-400X |
| Extended Depth of Focus (EDF) image can be constructed with NIS-Elements |
|
|
| |
Sample view under 5X objective with different zooms |
|
|
| |
|
 |
|
|
| |
| Configuration: Brightfield. Oblique Coherent Contrast (OCC) and Fluorescence |
| Magnification: 7.5X to 135X |
| Extended Depth of Focus (EDF) image can be constructed with NIS-Elements |
|
|
| |
EDF image of water plant constructed with Z-stack image |
Z-Stack |
EDF |
 |
 |
|
| |
|
|
| |
TS100: Cell Culture Microscope |
|
| |
|
 |
|
| |
|
|
Compact unit for long-term, time-lapse, live-cell imaging with
- Microscope
- Incubator
- High Sensitivity Camera
Configuration: Phase contrast + 2 Fluorescence
Application: Wound healing, Cell migration etc |
|
|
| |
|
|
Nikon Camera controller DS-L3 |
- Camera Controller
- Simple measurement
Distance between 2 points, length of perpendicular line, angle, diameter and circumference of circle, area of polygon etc.
- Touch Panel
* Please prepare thumbdrive as storing media |
|
| |
|
|
| |
Imaging System: NIS-Elements |
|
|
- Imaging software controls motorized microscope components, cameras and lasers
- 6D imaging capability
* Users should provide their own media to export generated data. |
| |
Image processing and analysis:
- Manual measurement
- Auto-measurement (binarization, object count)
- Time measurement
- Intensity measurement
- HDR image capture
- EDF
- Deconvolution
- Bio-analysis
- 3D video rendering etc
|
|
| |
